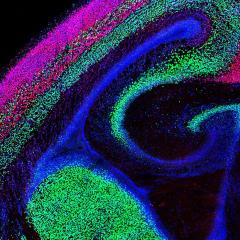
Professor Richards' laboratory investigates how the brain becomes wired up during development. The lab is focusing on the development of the cerebral cortex, a region of the brain where all higher order cognition is processed. The lab investigates the development of the largest fibre tract in the brain, called the corpus callosum, that connects neurons in the left and right cerebral hemispheres. Professor Richards' lab is interested in the development of the cortical midline and aspects of glial and neuronal development that impact the formation of midline structures such as the corpus callosum. The lab utilises both mouse and human tissue in its projects and applies the results to identifying the basis of agenesis of the corpus callosum, a condition that occurs in more than 300 different human congenital syndromes.
The Richards laboratory began a new era of research in 2015 by establishing protocols for examining brain wiring in human subjects. To launch this, the laboratory hosted an international conference called Cortical Connections, where a new international consortium of scientists and clinicians working on developmental brain disorders was established. The consortium will bring data together from different laboratories in Australia and internationally to help identify genes causing human brain disorders.
The consortium will also collect brain MRI data and neuropsychological data to try to understand the relationship between specific patterns of brain wiring and brain function. In this same area, the laboratory was awarded a National Institutes of Health grant with Professor Elliott Sherr (University of California, San Francisco) and Professor Bill Dobyns (Seattle University, Washington) who are internationally leading paediatric neurologists and geneticists, and a National Health and Medical Research Council grant with Professor Sherr and QBI researcher Associate Professor Tom Burne. These grants will specifically identify genes that cause human developmental brain disorders, and investigate their function.
For five years (2015-2020) Professor Richards was Deputy Director (Research) at The University of Queensland's Queensland Brain Institute. Professor Richards was appointed as an Officer (AO) of the Order of Australia (General Division) in 2019 for her "distinguished service to medical research and education in the field of developmental neurobiology, and to science and community engagement in science". She has been an NHMRC Principal Research Fellow since 2011. In 2015 she was elected as a Fellow of the Australian Academy of Science. In 2016 she was elected as a Fellow of the Australian Academy of Health and Medical Sciences and became President of the Australasian Neuroscience Society that year. Between 2017 and 2020 Professor Richards was Co-Chair of the Australian Brain Alliance and she was a member of the Federal Government's Million Minds Mission Advisory Panel from 2018 to 2020.
Group leader

Professor Linda Richards
Professorial Research Fellow(Group), Queensland Brain Institute
LinkedIn profile
irc5.org
ausdocc.org.au
+61 7 334 66492
richards@uq.edu.au
UQ Researcher Profile
Control of cellular differentiation in the developing brain
(2020-2022) ARC Discovery Project
Role of spontaneous activity in the formation of functional cortical circuits in vico
(2019-2021) NHMRC Project Grant
Normal and abnormal development of brain wiring and its impact on brain function
(2017-2021) NHMRC Principal Research Fellowship
Understanding cortical circuitry underlying sensory integration and the consequence of its developmental disruption
(2017–2020) NHMRC Project Grant
Neural circuits that mediate fear extinction
(2017-2021) NHMRC Project Grant
Astroglial remodelling of the interhemispheric midline is regulated by deleted in colorectal cancer (DCC) signalling and is required for corpus callosum formation
(2017–2019) NHMRC Project Grant
Unravelling the principles of bilateral brain wiring
(2016–2019) ARC Discovery Projects
Delayed radial glial maturation linked to NFI deficiency as an underlying cause of cortical defects in humans and mice
(2016–2018) NHMRC Project Grant
ACC: Callosal Agenesis as a Window into Common Neurodevelopmental Disorders (NIH grant administered by the University of California, San Francisco)
(2015–2017) Regents of the University of California
Targeting of callosal axons to duplicate cortical areas in the contralateral hemisphere Project Grant
(2014-2017) NHMRC Project Grant
Scholarly Book Chapters
Kozulin, P. and Richards L.J. (2016) Axonal Guidance: Making Connections. Neuroscience in the 21st Century: From basic to clinical (2nd edition), 201-224. Editors: Pfaff, D.W. and Volkow, N.D. Publisher: Springer New York.
Morcom L.R., Edwards T.J. and Richards L.J. (2015) Cortical architecture, midline guidance and tractography of 3-D white matter tracts. Axons and Brain Architecture, 289-313 Editor: Rockland K.S. Publisher: Elsevier.
Kozulin P., Almarza G., Gobius I. and Richards L.J. (2015) Investigating early formation of the cerebral cortex by in utero electroporation - methods and protocols. Prenatal and Postnatal Determinants of Brain Development - Recent Studies and Methodological Advances, 3-20. Editor: Walker D. Publisher: Springer Science+Business Media.
Chedotal A. and Richards L.J. (2010) Wiring the brain: The biology of neuronal guidance. Cold Spring Harbour Perspectives in Biology, 2:a001917. Editors: Tessier-Lavigne M. and Kolodkin A.L. Publisher: Cold Spring Harbour Laboratory Press.
Piper M., Dawson A., Lindwall C., Barry G., Plachez C. and Richards L.J. (2007) Emx and Nfi genes regulate cortical development and axon guidance in the telecephalon. Novartis Foundation Symposium 288, 230-245, final discussion 276-281. Editor: Bock G. and Goode J. Publisher: Wiley.
O'Leary D.D.M., Bastmeyer M., Daston M., Koester S.E., Richards L.J. and Yee K.T. (1996) Development of cortical output projections: axon guidance, target recognition, and plasticity. Integrative and Molecular Approach to Brain Function: Uehara Memorial Foundation Symposium-1996, 165-178. Editors: Ito M. and Miyashita Y. Publisher: Elsevier.
Bartlett P.F., Richards L.J., Kilpatrick T.J., Talman P.S., Bailey K., Brooker G., Dutton R., Koblar S., Nurcombe V., Ford M., et al. (1995) Factors Regulating the Differentiation of Neural Precursors in the Forebrain. CIBA Foundation Symposium 193 - Development of the Cerebral Cortex, 85-95. Editors: Blakemore, Cardew G. and Bock G.R. Publisher: Wiley.
Bartlett P.F., Drago J., Kilpatrick T.J., Richards L.J., Wookey P. and Murphy M. (1992) Regulation of the early development in the nervous system by growth factors. Development of the Central Nervous System in Vertebrates, 234: 1-21. Editors: Sharma S.C. and Goffinet A.M. Publisher: Springer US.
Refereed Journal Articles
Morcom L., Edwards T.J., Rider E., Jones-Davis D., Lim J.W.C., Chen K-S., Dean R., Bunt J., Ye Y., Gobius I., Suárez R., Mandelstam S., Sherr E.H. and Richards L.J. (2021) DRAXIN regulates interhemispheric fissure remodelling to influence the extent of corpus callosum formation eLife doi:10.7554/eLife.61618.
Morcom L., Gobius I., Marsh A.P.L., Suarez R., Lim J.W.C., Bridges C., Ye Y., Fenlon L.R., Zagar Y., Douglass A.M., Donahoo A-L.S., Fothergill T., Shaikh S., Kozulin P., Edwards T.J., Cooper H.M., IRC5 Consortium, Sherr E.H., Chedotal A., Leventer R.J., Lockhart P.J. and Richards L.J. (2021) DCC regulates astroglial development essential for telencephalic morphogenesis and corpus callosum formation eLife DOI:10.7554/eLife.61769. *Featured in Science editor’s choice in “Building bridges in the brain” feature https://science.sciencemag.org/content/372/6543/twil
Barker M.S., Knight J.L., Dean R., Mandelstam S., Richards L.J. and Robinson G.A. (2021) Verbal Adynamia and Conceptualization in Partial Rhombencephalosynapsis and Corpus Callosum Dysgenesis. Cognitive and Behavioral Neurology: March 2021 Volume 34 Issue 1 pp. 38-52 doi:10.1097/WNN.0000000000000261
The evolution, formation and connectivity of the anterior commissure
Fenlon L.R., Suarez R., Lynton Z. and Richards L.J. (2021) The evolution, formation and connectivity of the anterior commissure. Seminars in Cell and Developmental Biology https://doi.org/10.1016/j.semcdb.2021.04.009
NFIA and NFIB function as tumour suppressors in high-grade glioma in mice
Chen K-S., Lynton Z., Lim J.W.C., Robertson T., Gronostajski R.M., Bunt J. and Richards L.J. (2020) NFIA and NFIB function as tumour suppressors in high-grade glioma in mice Carcinogenesis, bgaa139, https://doi.org/10.1093/carcin/bgaa139.
Houston Z., Bunt J., Chen K.S., Puttick S., Howard C., Fletcher N., Fuchs A., Cui J., Ju Y., Cowin G.J., Song X., Boyd A., Mahler S., Richards LJ., Caruso F. and Thurecht K. (2020) Understanding the uptake of nanomedicines at different stages of brain cancer using a modular nanocarrier platform and precision bispecific antibodies. ACS Central Science 2020, 6, 5, 727–738, doi.org/10.1021/acscentsci.9b01299.
Pathogenic DDX3X mutations impair RNA metabolism and neurogenesis during fetal cortical development
Lennox A.L., Hoye M.L., Jiang R., Johnson-Kerner B.L., Suit L., Venkataramanan S., Sheehan C.J., Alsina F.C., Fregeau B., Aldinger K.A., Moey C., Lobach I., Alexandra A., Babovic-Vuksanovic D., Bézieau S., Blackburn P.R., Bunt J., Burglen L., Campeau P., Charles P., Chung B.H.Y., Cogné B., Curry C., D'Agostino M.D., DeBrosse S., Di Donato N., Faivre L., Héron D., Innes A.M., Isidor B., Keren B., Kimball A., Eric W. Klee E.W., Kuentz P., Küry S., Martin-Coignard D., Mirzaa G., Mignot C., Miyake N., Nava C., Nizon M., Rodriguez D., Snijders Blok L., Thauvin-Robinet C., Thevenon J., Vincent M., Ziegler A., Dobyns W., Richards L.J., Barkovich A.J., Floor S.N., Silver D.L., and Sherr E.H. (2020) Pathogenic DDX3X mutations impair RNA metabolism and neurogenesis during fetal cortical development. Neuron doi: 10.1016/j.neuron.2020.01.042.
Edwards T.J., Fenlon L.R., Dean R.J., Bunt J., Sherr E.H. and Richards L.J. (2020) Altered structural connectivity networks in a mouse model of complete and partial dysgenesis of the corpus callosum Neuroimage doi.org/10.1016/j.neuroimage.2020.116868.
Paolino A., Fenlon L.R., Kozulin P., Haines E., Lim J.W.C., Richards L.J. and Suárez R. (2020) Differential timing of a conserved transcriptional network underlies divergent cortical projection routes across mammalian evolution. Proc. Natl. Acad. Sci. USA Sciences doi.org/10.1073/pnas.1922422117.
Spencer-Smith M., Knight J.L., Lacaze E., IRC5 Consortium, Depienne C., Lockhart P.J., Richards L.J., Heron D., Leventer R.J. and Robinson G.A. (2020) Callosal agenesis and congenital mirror movements: clinical, imaging and neuropsychological outcomes associated with DCC mutations Developmental Medicine & Child Neurology doi.org/10.1111/dmcn.14486.
Variants in Nuclear Factor I Genes Influence Growth and Development
Zenker M., Bunt J., Schanze I., Schanze D., Piper M., Priolo M., Gerkes E.H., Gronostajski R.M., Richards L.J., Vogt J., Wessels M.W., Hennekam R.C. (2019) Variants in Nuclear Factor I Genes Influence Growth and Development. American Journal of Medical Genetics Part C: Seminars in Medical Genetics doi:10.1002/ajmg.c.31747.
Pajtler K., Wei Y., Okonechnikov K., Benites P., Vouri M., Zhang L., Brabetz S., Sieber L., Gulley M., Mauermann M., Wedig T., Mack N., Kawasawa Y., Sharma T., Zuckermann M., Andreiuolo F., Holland E., Maass K., Körkel-Qu H., Liu H-K., Sahm F., Capper D., Bunt J., Richards L.J., Jones D., Korshunov A., Chavez L., Lichter P., Hoshino M., Pfister S., Kool M., Li W. and Kawauchi D. (2019) YAP1 subgroup supratentorial ependymoma requires TEAD and Nuclear factor I-mediated transcriptional programmes for tumourigenesis. Nature Communications doi.org/10.1038/s41467-019-11884-5.
Transcription factors NFIA and NFIB induce cellular differentiation high-grade astrocytoma
Chen K-S., Bunt J., Bridges C.R., Lynton Z., Lim J.W.C., Stringer B.W., Rajagopal R., Wong K-T., Ganesan D., Ariffin H., Day B.W. and Richards L.J. (2019) Transcription factors NFIA and NFIB induce cellular differentiation high-grade astrocytoma. Journal of Neuro-Oncology doi:10.1007/s11060-019-03352-3.
Shared and differential features of Robo3 expression pattern in amniotes
Friocourt F., Kozulin, P., Belle M., Suárez R., Di-Poï N., Richards L.J., Giacobini P. and Chédotal C. (2019). Shared and differential features of Robo3 expression pattern in amniotes. The Journal of Comparative Neuroloy doi: 10.1002/cne.24648.
A neuroethics framework for the Australian Brain Initiative
Richards L.J. (corresponding author) Australian Brain Alliance (2019) A neuroethics framework for the Australian Brain Initiative. Neuron doi: 10.1016/j.neuron.2019.01.004.
Hearne, L.J., Dean, R., Robinson, G., Richards L.J., Mattingley J. and Cocchi L. (2018) Increased cognitive complexity reveals abnormal brain network activity in individuals with corpus callosum dysgenesis. Neuroimage doi.org/10.1016/j.nicl.2018.11.005.
NFIB haploinsufficiency is associated with intellectual disability and marcocephaly
Schanze I., Bunt J., Boppudi S., Lim J.W.C., Schanze D., Dean R.J., Alders M., Blanchet P., Attié-Bitach T., Berland S., Bridges C.J., Cho M.T., Desai S., Dobyns W.B., Donnai D., Douglas J., Earl D.L., Edwards T.J., Faivre L., Genevieve D., Gérard M., Gatinois V., Holder-Espinasse M., Huth S.F., Izumi, K., Kerr, B., Lacaze E., Lakeman P., Mirzaa G., Morgan S., Nowak C., Peeters H., Petit F., Pilz D.T., Puechberty J., Reinstein E., Rivière J-B., Santani A.B., Schneider A., Sherr E.H., Smith-Hicks C., Wieland I., Zackai E., , Gronostajski R.M., Zenker M., and Richards L.J. (2018) NFIB haploinsufficiency is associated with intellectual disability and marcocephaly. The American Journal of Human Genetics doi: 10.1016/j.ajhg.2018.10.006.
Suárez R., Paolino A., Fenlon L.R., Morcom L.R., Kozulin P., Kurniawan N.D., and Richards L.J. (2018) A pan-mammalian map of interhemispheric brain connections predates the evolution of the corpus callosum. Proceedings of the National Academy of Sciences doi.org/10.1073/pnas.1808262115.
Edwards T.J., Marsh A.P.L., Lockhart P.J., Richards L.J. and Leventer R.J. (2018) Teaching Neuroimages: Imaging features of DCC-mediated mirror movements and isolated agenesis of the corpus callosum. Neurology doi.org/10.1212/WNL.0000000000006085.
Transcriptional control of long-range cortical projections.
Paolino A., Fenlon L.R., Suárez R. and Richards, L.J. (2018) Transcriptional control of long-range cortical projections. Current Opinion in Neurobiology 53:57-65 doi.org/10.1016/j.conb.2018.05.005.
DCC is required for the development of nociceptive topognosis in mice and humans
Da Silva R.V., Johannssen H.C., Wyss M.T., Roome R.B., Bourojeni F.B., Stifani N., Marsh A.P.L., Ryan M.M., Lockhart P.J., Leventer R.J., Richards, L.J., Rosenblatt B., Srour M., Weber B., Zeilhofer, H.U. and Kania A. (2018) DCC is required for the development of nociceptive topognosis in mice and humans. Cell Reports 22:1105-1114.
Pharmacogenetic stimulation of neuronal activity increases myelination in an axon-specific manner
Mitew S., Gobius I., Fenlon L., McDougall S., Hawkes D., Xing Y., Bujalka H., Gundlach A., Richards L.J., Kilpatrick T.J., Merson T. and Emery B. (2017) Pharmacogenetic stimulation of neuronal activity increases myelination in an axon-specific manner. Nature Comm. 10.1038/s41467-017-02719-2.
Marsh A.P.L., Edwards T.J., Galea C., Cooper H.M., Engle E.C., Jamuar S.S., Méneret A., Moutard M-L., Nava C., Rastetter A., Robinson G., Rouleau G., Roze E., Spencer-Smith M., Trouillard O., Billette de Villemeur T., Walsh C.A., Yu T.W., IRC5 Consortium, Heron D., Sherr E.H., Richards L.J., Depienne C., Leventer R.J. and Lockhart P.J. (2017) DCC mutation update: congenital mirror movements, isolated agenesis of the corpus callosum and developmental split brain syndrome. Human Mutation 2018 Jan;39(1):23-39. doi: 10.1002/humu.23361.
Matuzelski E., Bunt J., Harkins D., Lim J.W.C., Gronostajski R.M., Richards L.J., Harris L. and Piper M. (2017) Transcriptional regulation of Nfix by NFIB drives astrocytic maturation within the developing spinal cord. Developmental Biology 2017 Dec 15;432(2):286-297. doi: 10.1016/j.ydbio.2017.10.019.
The convergent roles of the nuclear factor I transcription factors in development and cancer
Chen K-S., Lim J.W.C., Richards L.J. and Bunt J. (2017) The convergent roles of the nuclear factor I transcription factors in development and cancer. Cancer Letters 2017 Dec 1:410:124-138. Doi 10.1016/j.canlet.2017.09.015.
Combined allelic dosage of Nfia and Nfib regulates cortical development.
Bunt J., Osinski J.M., Lim J.W.C., Vidovic D., Ye Y., Zalucki O., O’Connor T.R., Harris L., Gronostajski R.M., Richards L.J., and Piper M. (2017) Combined allelic dosage of Nfia and Nfib regulates cortical development. Brain and Neuroscience Advances Volume 1: 1–21.
Paolino A., Fenlon L.R., Kozulin P., Richards L.J. and Suarez R. (2017) Multiple events of gene manipulation via in pouch electroporation in a marsupial model of mammalian forebrain development. Journal of Neuroscience Methods Volume 293: 45-52 doi: 10.1016/j.jneumeth.2017.09.004.
Suarez R., Paolino A., Kozulin P., Fenlon L.R., Morcom L.R., Englebright R., O’Hara P.J., Murray P.J. and Richards L.J. (2017) Development of body, head and brain features in the Australian fat-tailed dunnart (Sminthopsis crassicaudata; Marsupialia: Dasyuridae); A postnatal model of forebrain formation. PLOS ONE 12(9): e0184450.
Gobius I., Suarez R., Morcom L., Paolino A., Edwards T.J., Kozulin P. and Richards L.J. (2017) Astroglial-mediated remodeling of the interhemispheric midline during telencephalic development is exclusive to eutherian mammals. Neural Development 12:9 DOI: 10.1186/s13064-017-0086-1.
Chen K-S., Harris L., Lim J.W.C., Harvey T., Piper M., Gronostajski R., Richards L.J. and Bunt J. (2017) Differential neuronal and glial expression of Nuclear factor I proteins in the dorsal forebrain of adult mice. Journal of Comparative Neurology DOI: 10.1002/cne.24206.
Reducing the burden of neurological disease and mental illness
Double K. and Richards L.J. (corresponding authors) (2017) Reducing the burden of neurological disease and mental illness. Medical Journal of Australia 206 (8): 341-342.
Mutations in DCC cause isolated agenesis of the corpus callosum with incomplete penetrance
Marsh A.P.L., Herron D., Edwards T.J., et al, Richards L.J.§ Lockhart P.J.§ and Depienne C.§ (2017) Mutations in DCC cause isolated agenesis of the corpus callosum with incomplete penetrance. Nature Genetics (doi:10.1038/ng.3794). §Equal last and senior authors.
Fenlon L.R., Suarez, R. and Richards L.J. (2017) The anatomy, organisation and development of contralateral callosal projections of the mouse somatosensory cortex. Brain and Neuroscience Advances 10.1177/2398212817694888.
Transcriptional regulation of intermediate progenitor cell generation during hippocampal development
Harris, Lachlan, Zalucki, Oressia, Gobius, Ilan, McDonald, Hannah, Osinki, Jason, Harvey, Tracey J., Essebier, Alexandra, Vidovic, Diana, Gladwyn-Ng, Ivan, Burne, Thomas H., Heng, Julian I., Richards, Linda J., Gronostajski, Richard M. and Piper, Michael (2016) Transcriptional regulation of intermediate progenitor cell generation during hippocampal development. Development, 143 24: 4620-4630. doi:10.1242/dev.140681
Gobius, Ilan, Morcom, Laura, Sua´rez, Rodrigo, Bunt, Jens, Bukshpun, Polina, Reardon, William, Dobyns, William B., Rubenstein, John L.R., Barkovich, James, Sherr, Elliott H. and Richards, Linda J. (2016) Astroglial-mediated remodeling of the interhemispheric midline is required for the formation of the corpus callosum. Cell Reports, 17 3: 735-747. doi:10.1016/j.celrep.2016.09.033
Altered structural connectome in adolescent socially isolated mice
Liu, Cirong, Li, Yonghui, Edwards, Timothy J., Kurniawan, Nyoman D., Richards, Linda J. and Jiang, Tianzi (2016) Altered structural connectome in adolescent socially isolated mice. Neuroimage, 139 259-270. doi:10.1016/j.neuroimage.2016.06.037
Edwards, Timothy J., Sherr, Elliott H., Barkovich, A. James and Richards, Linda J. (2016) Reply: ARID 1B mutations are the major genetic cause of corpus callosum anomalies in patients with intellectual disability. Brain, 139 11: e65. doi:10.1093/brain/aww171
Nuclear factor one B (NFIB) encodes a subtype-specific tumour suppressor in glioblastoma
Stringer, Brett W., Bunt, Jens, Day, Bryan W., Barry, Guy, Jamieson, Paul R., Ensbey, Kathleen S., Bruce, Zara C., Goasdoué, Kate, Vidal, Helene, Charmsaz, Sara, Smith, Fiona M., Cooper, Leanne T., Piper, Michael, Boyd, Andrew W. and Richards, Linda J. (2016) Nuclear factor one B (NFIB) encodes a subtype-specific tumour suppressor in glioblastoma. Oncotarget, 7 20: 29306-12320. doi:10.18632/oncotarget.8720
NFIX regulates proliferation and migration within the murine SVZ neurogenic niche
Heng, Yee Hsieh Evelyn, Zhou, Bo, Harris, Lachlan, Harvey, Tracey, Smith, Aaron, Horne, Elise, Martynoga, Ben, Andersen, Jimena, Achimastou, Angeliki, Cato, Kathleen, Richards, Linda J., Gronostajski, Richard M., Yeo, Giles S., Guillemot, François, Bailey, Timothy L. and Piper, Michael (2015) NFIX regulates proliferation and migration within the murine SVZ neurogenic niche. Cerebral Cortex, 25 10: 3758-3778. doi:10.1093/cercor/bhu253
MicroRNA-153 regulates the acquisition of gliogenic competence by neural stem cells
Tsuyama, Jun, Bunt, Jens, Richards, Linda J., Iwanari, Hiroko, Mochizuki, Yasuhiro, Hamakubo, Takao, Shimazaki, Takuya and Okano, Hideyuki (2015) MicroRNA-153 regulates the acquisition of gliogenic competence by neural stem cells. Stem Cell Reports, 5 3: 365-377. doi:10.1016/j.stemcr.2015.06.006
EMX1 regulates NRP1-mediated wiring of the mouse anterior cingulate cortex
Lim, Jonathan W. C., Donahoo, Amber-Lee S., Bunt, Jens, Edwards, Timothy J., Fenlon, Laura R., Liu, Ying, Zhou, Jing, Moldrich, Randal X., Piper, Michael, Gobius, Ilan, Bailey, Timothy L., Wray, Naomi R., Kessaris, Nicoletta, Poo, Mu-Ming, Rubenstein, John L. R. and Richards, Linda J. (2015) EMX1 regulates NRP1-mediated wiring of the mouse anterior cingulate cortex. Development, 142 21: 3746-3757. doi:10.1242/dev.119909
Fenlon, Laura R., Liu, Sha, Gobius, Ilan, Kurniawan, Nyoman D., Murphy, Skyle, Moldrich, Randal X. and Richards, Linda J. (2015) Formation of functional areas in the cerebral cortex is disrupted in a mouse model of autism spectrum disorder. Neural Development, 10 10: 1-14. doi:10.1186/s13064-015-0033-y
Contralateral targeting of the corpus callosum in normal and pathological brain function
Fenlon, Laura R. and Richards, Linda J. (2015) Contralateral targeting of the corpus callosum in normal and pathological brain function. Trends in Neurosciences, 38 5: 264-272. doi:10.1016/j.tins.2015.02.007
PAX6 does not regulate Nfia and Nfib expression during neocortical development
Bunt, Jens, Lim, Jonathan W., Zhao, Lu, Mason, Sharon and Richards, Linda J. (2015) PAX6 does not regulate Nfia and Nfib expression during neocortical development. Scientific Reports, 5 Art No.: 10668: . doi:10.1038/srep10668
Mapk/Erk activation in an animal model of social deficits shows a possible link to autism
Faridar, Alireza, Jones-Davis, Dorothy, Rider, Eric, Li, Jiang, Gobius, Ilan, Morcom, Laura, Richards, Linda, Sen, Saunak and Sherr, Elliott (2014) Mapk/Erk activation in an animal model of social deficits shows a possible link to autism. Molecular Autism, 5 57: 1-12. doi:10.1186/2040-2392-5-57
Schmidt, Ewoud Roberto Eduard, Brignani, Sara, Adolfs, Youri, Lemstra, Suzanne, Demmers, Jeroen, Vidaki, Marina, Donahoo, Amber-Lee Skye, Lillevali, Kersti, Vasar, Eero, Richards, Linda Jane, Karagogeos, Domna, Kolk, Sharon Margriet and Pasterkamp, Ronald Jeroen (2014) Subdomain-mediated axon-axon signaling and chemoattraction cooperate to regulate afferent innervation of the lateral habenula. Neuron, 83 2: 372-387. doi:10.1016/j.neuron.2014.05.036
Evolution and development of interhemispheric connections in the vertebrate forebrain
Suarez, Rodrigo, Gobius, Ilan and Richards, Linda J. (2014) Evolution and development of interhemispheric connections in the vertebrate forebrain. Frontiers in Human Neuroscience, 8 JULY: 1-14. doi:10.3389/fnhum.2014.00497
Balanced interhemispheric cortical activity is required for correct targeting of the corpus callosum
Suarez, Rodrigo, Fenlon, Laura R., Marek, Roger, Avitan, Lilach A, Sah, Pankaj, Goodhill, Geoffrey J. and Richards, Linda J. (2014) Balanced interhemispheric cortical activity is required for correct targeting of the corpus callosum.Neuron, 82 6: 1289-1298. doi:10.1016/j.neuron.2014.04.040
Unc5C and DCC act downstream of Ctip2 and Satb2 and contribute to corpus callosum formation
Srivatsa, Swathi, Parthasarathy, Srinivas, Britanova, Olga, Bormuth, Ingo, Donahoo, Amber-Lee, Ackerman, Susan L., Richards, Linda J. and Tarabykin, Victor (2014) Unc5C and DCC act downstream of Ctip2 and Satb2 and contribute to corpus callosum formation. Nature Communications, 5 . doi:10.1038/ncomms4708
NFIB-mediated repression of the epigenetic factor Ezh2 regulates cortical development
Piper, Michael, Barry, Guy, Harvey, Tracey J., McLeay, Robert, Smith, Aaron G., Harris, Lachlan, Mason, Sharon, Stringer, Brett W., Day, Bryan W., Wray, Naomi R., Gronostajski, Richard M., Bailey, Timothy L., Boyd, Andrew W. and Richards, Linda J. (2014) NFIB-mediated repression of the epigenetic factor Ezh2 regulates cortical development.Journal of Neuroscience, 34 8: 2921-2930. doi:10.1523/JNEUROSCI.2319-13.2014
Diffusion MR microscopy of cortical development in the mouse embryo
Aggarwal, Manisha, Gobius, Ilan, Richards, Linda J. and Mori, Susumu (2014) Diffusion MR microscopy of cortical development in the mouse embryo. Cerebral Cortex, 25 7: 1970-1980. doi:10.1093/cercor/bhu006
Wnt5a induces Ryk-dependent and -independent effects on callosal axon and dendrite growth
Clark, Charlotte E. J., Richards, , Linda J., Stacker, Steven A. and Cooper, Helen M. (2014) Wnt5a induces Ryk-dependent and -independent effects on callosal axon and dendrite growth. Growth Factors, 32 1: 11-17. doi:10.3109/08977194.2013.875544
Clinical, genetic and imaging findings identify new causes for corpus callosum development syndromes
Edwards, Timothy J., Sherr, Elliott H., Barkovich, A. James and Richards, Linda J. (2014) Clinical, genetic and imaging findings identify new causes for corpus callosum development syndromes. Brain, 137 6: 1579-1613. doi:10.1093/brain/awt358
NFIX regulates neural progenitor cell differentiation during hippocampal morphogenesis
Heng, Yee Hsieh Evelyn, McLeay, Robert C., Harvey, Tracey J., Smith, Aaron G., Barry, Guy, Cato, Kathleen, Plachez, Celine, Little, Erica, Mason, Sharon, Dixon, Chantelle, Gronostajski, Richard M., Bailey, Timothy L., Richards, Linda J. and Piper, Michael (2014) NFIX regulates neural progenitor cell differentiation during hippocampal morphogenesis.Cerebral Cortex, 24 1: 261-279. doi:10.1093/cercor/bhs307
Fothergill, Thomas, Donahoo, Amber-Lee S., Douglass, Amelia, Zalucki, Oressia, Yuan, Jiajia, Shu, Tianzhi, Goodhill, Geoffrey J. and Richards, Linda J. (2014) Netrin-DCC signaling regulates corpus callosum formation through attraction of pioneering axons and by modulating Slit2-mediated repulsion. Cerebral Cortex, 24 5: 1138-1151. doi:10.1093/cercor/bhs395
X-linked microtubule-associated protein, Mid1, regulates axon development
Lu, Tingjia, Chen, Renchao, Cox, Timothy C., Moldrich, Randal X., Kurniawan, Nyoman, Tan, Guohe, Perry, Jo K., Ashworth, Alan, Bartlett, Perry F., Xu, Li, Zhang, Jing, Lu, Bin, Wu, Mingyue, Shen, Qi, Liu, Yuanyuan, Richards, Linda J. and Xiong, Zhiqi (2013) X-linked microtubule-associated protein, Mid1, regulates axon development.Proceedings of the National Academy of Sciences of the United States of America, 110 47: 19131-19136. doi:10.1073/pnas.1303687110
Huang, Hao, Jeon, Tina, Sedmak, Goran, Pletikos, Mihovil, Vasung, Lana, Xu, Xuming, Yarowsky, Paul, Richards, Linda J., Kostović, Ivica, Šestan, Nenad and Mori, Susumu (2013) Coupling diffusion imaging with histological and gene expression analysis to examine the dynamics of cortical areas across the fetal period of human brain development. Cerebral Cortex, 23 11: 2620-2631. doi:10.1093/cercor/bhs241
Axon position within the corpus callosum determines contralateral cortical projection
Zhou, Jing, Wen, Yunqing, She, Liang, Sui, Ya-nan, Liu, Lu, Richards, Linda J. and Poo, Mu-ming (2013) Axon position within the corpus callosum determines contralateral cortical projection. Proceedings of the National Academy of Sciences of the United States of America, 110 29: E2714-E2723. doi:10.1073/pnas.1310233110
Heterozygosity for Nuclear Factor One X Affects Hippocampal-Dependent Behaviour in Mice
Harris, Lachlan, Dixon, Chantelle, Cato, Kathleen, Heng, Yee Hsieh Evelyn, Kurniawan, Nyoman D., Ullmann, Jeremy F. P., Janke, Andrew L., Gronostajski, Richard M., Richards, Linda J., Burne, Thomas H. J. and Piper, Michael (2013) Heterozygosity for Nuclear Factor One X Affects Hippocampal-Dependent Behaviour in Mice. PLoS ONE, 8 6: . doi:10.1371/journal.pone.0065478
Loss of Usp9x disrupts cortical architecture, hippocampal development and TGFβ-mediated axonogenesis
Stegeman, Shane, Jolly, Lachlan A., Premarathne, Susitha, Gecz, Jozef, Richards, Linda J., Mackay-Sim, Alan and Wood, Stephen A.. (2013) Loss of Usp9x disrupts cortical architecture, hippocampal development and TGFβ-mediated axonogenesis. PLoS ONE, 8 7: e68287.1-e68287.11. doi:10.1371/journal.pone.0068287
Expression of nuclear factor one A and -B in the olfactory bulb
Plachez, Celine, Cato, Kathleen, McLeay, Robert C., Heng, Yee Hsieh Evelyn, Bailey, Timothy L., Gronostajski, Richard M., Richards, Linda J., Puche, Adam C. and Piper, Michael (2012) Expression of nuclear factor one A and -B in the olfactory bulb. The Journal of Comparative Neurology, 520 14: 3135-3149. doi:10.1002/cne.23081
Multiple Slits regulate the development of midline glial populations and the corpus callosum
Unni, Divya K., Piper, Michael, Moldrich, Randal X., Gobius, Ilan, Liu, Sha, Fothergill, Thomas, Donahoo, Amber-Lee S., Baisden, John M., Cooper, Helen M. and Richards, Linda J. (2012) Multiple Slits regulate the development of midline glial populations and the corpus callosum. Developmental Biology, 365 1: 36-49. doi:10.1016/j.ydbio.2012.02.004
Nuclear factor I genes regulate neuronal migration
Heng, Yee Hsieh Evelyn, Barry, Guy, Richards, Linda J. and Piper, Michael (2012) Nuclear factor I genes regulate neuronal migration. NeuroSignals, 20 3: 159-167. doi:10.1159/000330651
Shekarabi, Masoud, Moldrich, Randal X., Rasheed, Sarah, Salin-Cantegrel, Adele, Laganiere, Janet, Rochefort, Daniel, Hince, Pascale, Huot, Karine, Gaudet, Rebecca, Kurniawan, Nyoman, Sotocinal, Susana G., Ritchie, Jennifer, Dion, Patrick A., Mogil, Jeffrey S., Richards, Linda J. and Rouleau, Guy A. (2012) Loss of neuronal potassium/chloride cotransporter 3 (KCC3) is responsible for the degenerative phenotype in a conditional mouse model of hereditary motor and sensory neuropathy associated with agenesis of the corpus callosum. Journal of Neuroscience, 32 11: 3865-3876. doi:10.1523/JNEUROSCI.3679-11.2012
Piper, Michael, Harris, Lachlan, Barry, Guy, Heng, Yee Hsieh Evelyn, Plachez, Celine, Gronostajski, Richard M. and Richards, Linda J. (2011) Nuclear factor one X regulates the development of multiple cellular populations in the postnatal cerebellum. The Journal of Comparative Neurology, 519 17: 3532-3548. doi:10.1002/cne.22721
Subramanian, Lakshmi, Sarkar, Anindita, Shetty, Ashwin S., Muralidharan, Bhavana, Padmanabhan, Hari, Piper, Michael, Monuki, Edwin S., Bach, Ingolf, Gronostajski, Richard M., Richards, Linda J. and Tole, Shubha (2011) Transcription factor Lhx2 is necessary and sufficient to suppress astrogliogenesis and promote neurogenesis in the developing hippocampus. Proceedings of the National Academy of Sciences of the United States of America, 108 27: E-265-E-274. doi:10.1073/pnas.1101109108
Fgfr3 regulates development of the caudal telencephalon
Moldrich, Randal X., Mezzera, Cecilia, Holmes, William M., Goda, Sailaja, Brookfield, Sam J., Rankin, Alastair J., Barr, Emily, Kurniawan, Nyoman, Dewar, Deborah, Richards, Linda J., López-Bendito, Guillermina and Iwata, Tomoko (2011) Fgfr3 regulates development of the caudal telencephalon. Developmental Dynamics, 240 6: 1586-1599. doi:10.1002/dvdy.22636
Cyclic nucleotide-dependent switching of mammalian axon guidance depends on gradient steepness
Thompson, Andrew W., Pujic, Zac, Richards, Linda J. and Goodhill, Geoffrey J. (2011) Cyclic nucleotide-dependent switching of mammalian axon guidance depends on gradient steepness. Molecular and Cellular Neuroscience, 47 1: 45-52. doi:10.1016/j.mcn.2011.02.012
An MRI-based atlas and database of the developing mouse brain
Chuang, Nelson, Mori, Susumu, Yamamoto, Akira, Jiang, Hangyi, Ye, Xin, Xu, Xin, Richards, Linda J., Nathans, Jeremy, Miller, Michael I., Toga, Arthur W., Sidman, Richard L. and Zhang, Jiangyang (2011) An MRI-based atlas and database of the developing mouse brain. NeuroImage, 54 1: 80-89. doi:10.1016/j.neuroimage.2010.07.043
Molecular regulation of the developing commissural plate
Moldrich, Randal X., Gobius, Ilan, Pollak, Thomas, Zhang, Jiangyang, Ren, Tianbo, Brown, Lucia, Mori, Susumu, Romero, Camino De Juan, Britanova, Olga, Tarabykin, Victor and Richards, Linda J. (2010) Molecular regulation of the developing commissural plate. Journal of Comparative Neurology, 518 18: 3645-3661. doi:10.1002/cne.22445
Comparative mouse brain tractography of diffusion magnetic resonance imaging
Moldrich, Randal X., Pannek, Kerstin, Hoch, Renee, Rubenstein, John L., Kurniawan, Nyoman D. and Richards, Linda J. (2010) Comparative mouse brain tractography of diffusion magnetic resonance imaging. NeuroImage, 51 3: 1027-1036. doi:10.1016/j.neuroimage.2010.03.035
Piper, Michael, Barry, Guy, Hawkins, John, Mason, Sharon, Lindwall, Charlotta, Little, Erica, Sarkar, Anindita, Smith, Aaron G., Moldrich, Randal X., Boyle, Glen M., Tole, Shubjha, Gronostajski, Richard M., Bailey, Timothy L. and Richards, Linda J. (2010) NFIA controls telencephalic progenitor cell differentiation through repression of the Notch effector Hes1. Journal of Neuroscience, 30 27: 9127-9139. doi:10.1523/JNEUROSCI.6167-09.2010
Wiring the brain: The biology of neuronal guidance
Chedotal, Alain and Richards, Linda J. (2010) Wiring the brain: The biology of neuronal guidance. Cold Spring Harbor Perspectives in Biology, 2 6: a001917-1-a001917-17. doi:10.1101/cshperspect.a001917
Multiple non-cell-autonomous defects underlie neocortical callosal dysgenesis in Nfib-deficient mice
Piper, Michael, Moldrich, Randal, Lindwall, Charlotta, Little, Erica, Barry, Guy, Mason, Sharon, Sunn, Nana, Kurniawan, Nyoman D., Gronostajski, Richard M. and Richards, Linda J. (2009) Multiple non-cell-autonomous defects underlie neocortical callosal dysgenesis in Nfib-deficient mice. Neural Development, 4 Article # 43: 1-16. doi:10.1186/1749-8104-4-43
Understanding the mechanisms of callosal development through the use of transgenic mouse models
Donahoo, Amber-Lee and Richards, Linda J. (2009) Understanding the mechanisms of callosal development through the use of transgenic mouse models. Seminars in Pediatric Neurology, 16 3: 127-142. doi:10.1016/j.spen.2009.07.003
Piper, Michael, Plachez, Celine, Zalucki, Oressia, Fothergill, Thomas, Goudreau, Guy, Erzurumlu, Reha, Gu, Chenghua and Richards, Linda J. (2009) Neuropilin 1-Sema signaling regulates crossing of cingulate pioneering axons during development of the corpus callosum. Cerebral Cortex, 19 Supplement 1: i11-i21. doi:10.1093/cercor/bhp027
A Bayesian model predicts the response of axons to molecular gradients
Mortimer, Duncan, Feldner, Julia, Vaughan, Timothy, Vetter, Irina, Pujic, Zac, Rosoff, William J., Burrage, Kevin, Dayan, Peter, Richards, Linda J. and Goodhill, Geoffrey J. (2009) A Bayesian model predicts the response of axons to molecular gradients. Proceedings of the National Academy of Sciences of the United States of America, 106 25: 10296-10301. doi:10.1073/pnas.0900715106
Why schizophrenia epidemiology needs neurobiology - And vice versa
McGrath, John J. and Richards, Linda J. (2009) Why schizophrenia epidemiology needs neurobiology - And vice versa.Schizophrenia Bulletin, 35 3: 577-581. doi:10.1093/schbul/sbp004
Huang, Hao, Xue, Rong, Zhang, Jiangyang, Ren, Tianbo, Richards, Linda J., Yarowsky, Paul, Miller, Michael I. and Mori, Susumu (2009) Anatomical characterization of human fetal brain development with diffusion tensor magnetic resonance imaging. The Journal of Neuroscience, 29 13: 4263-4273. doi:10.1523/JNEUROSCI.2769-08.2009
Kumbasar, Asli, Plachez, Celine, Gronostajski, Richard M. and Richards, Linda J. (2009) Absence of the Transcription Factor Nfib Delays the Formation of the Basilar Pontine and Other Mossy Fiber Nuclei. The Journal of Comparative Neurology, 513 1: 98-112. doi:10.1002/cne.21943
Nuclear Factor One Transcription Factors in CNS Development
Mason, Sharon, Piper, Michael, Gronostajski, Richard M. and Richards, Linda J. (2009) Nuclear Factor One Transcription Factors in CNS Development. Molecular Neurobiology, 39 1: 10-23. doi:10.1007/s12035-008-8048-6
Specific glial populations regulate hippocampal morphogenesis
Barry, Guy, Michael Piper, Lindwall, Charlotta, Moldrich, Randal, Mason, Sharon, Little, Erica, Sarkar, Anindita, Tole, Shubha, Gronostajski, Richard M. and Richards, Linda J. (2008) Specific glial populations regulate hippocampal morphogenesis. The Journal of Neuroscience, 28 47: 12328-12340. doi:10.1523/JNEUROSCI.4000-08.2008
The transcription factor Nfix is essential for normal brain development
Campbell, Christine E., Piper, Michael, Plachez, Celine, Yeh, Yu-Ting, Baizer, Joan S., Osinski, Jason M., Litwack, E David, Richards, Linda J. and Gronostajski, Richard M. (2008) The transcription factor Nfix is essential for normal brain development. BMC Developmental Biology, 8 52: 1-18. doi:10.1186/1471-213X-8-52
Mortimer, Duncan, Fothergill, Thomas, Pujic, Zac, Richards, Linda J. and Goodhill, Geoffrey J. (2008) Growth cone chemotaxis. Trends in Neurosciences, 31 2: 90-98. doi:10.1016/j.tins.2007.11.008
Nuclear Factor I Gene Expression in the Developing Forebrain
Plachez, Celine, Lindwall, Charlotta, Sunn, Nana, Piper, Michael, Moldrich, Randal X., Campbell, Christine E., Osinski, Jason M., Gronostajski, Richard M. and Richards, Linda J. (2008) Nuclear Factor I Gene Expression in the Developing Forebrain. Journal of Comparative Neurology, 508 3: 385-401. doi:10.1002/cne.21645
Plachez, Céline, Andrews, William, Liapi, Anastasia, Knoell, Bernd, Drescher, Uwe, Mankoo, Baljinder, Zhe, Liu, Mambetisaeva, Elvira, Annan, Adelaide, Bannister, Lawrence, Parnavelas, John G., Richards, Linda J. and Sundaresan, Vasi (2008) Robos are required for the correct targeting of retinal ganglion cell axons in the visual pathway of the brain. Molecular and Cellular Neuroscience, 37 4: 719-730. doi:10.1016/j.mcn.2007.12.017
Agenesis of the corpus callosum: genetic, developmental and functional aspects of connectivity
Paul, L. K., Brown, W. S., Adolphs, R., Tyszka J. M., Richards, L. J., Mukherjee, P. and Sherr, E. H. (2007) Agenesis of the corpus callosum: genetic, developmental and functional aspects of connectivity. Nature Reviews. Neuroscience,8 4: 287-299. doi:10.1038/nrn2107
Commissure formation in the mammalian forebrain
Lindwall, Charlotta, Fothergill, Thomas and Richards, Linda J. (2007) Commissure formation in the mammalian forebrain. Current Opinion In Neurobiology, 17 1: 3-14. doi:10.1016/j.conb.2007.01.008
Ren, T, Zhang, JY, Plachez, C, Mori, S and Richards, LJ (2007) Diffusion tensor magnetic resonance imaging and tract-tracing analysis of probst bundle structure in Netrin1- and DCC-deficient mice. Journal of Neuroscience, 27 39: 10345-10349. doi:10.1523/JNEUROSCI.2787-07.2007
White and gray matter development in human fetal, newborn and pediatric brains
Huang, Hao, Zhang, Jiangyang, Wakana, Setsu, Zheng, Weihong, Ren,Tianbo, Richards, Linda J., Yarowsky, Paul, Donohue, Pamela, Graham, Ernest, Van Zijl, Peter C.M. and Mori, Susumu (2006) White and gray matter development in human fetal, newborn and pediatric brains. Neuroimage, 33 1: 27-38. doi:10.1016/j.neuroimage.2006.06.009
Identification of candidate genes at the corticoseptal boundary during development
Shen, Wei-Bin, Plachez, Celine, Mongi, Alka S. and Richards, Linda J. (2006) Identification of candidate genes at the corticoseptal boundary during development. Gene Expression Patterns, 6 5: 471-481. doi:10.1016/j.modgep.2005.11.004
Robo1 regulates the development of major axon tracts and interneuron migration in the forebrain
Andrews, W., Liapi, A., Plachez, C., Camurri, L., Zhang, J. Y., Mori, S., Murakami, F., Parnavelas, J. G., Sundaresan, V. and Richards, L. J. (2006) Robo1 regulates the development of major axon tracts and interneuron migration in the forebrain. Development, 133 11: 2243-2252. doi:10.1242/dev.02379
Ren, T. B., Anderson, A., Shen, W. B., Huang, H., Plachez, C., Zhang, J. Y., Mori, S., Kinsman, S. L. and Richards, L. J. (2006) Imaging, anatomical, and molecular analysis of callosal formation in the developing human fetal brain.Anatomical Record - Part A: Discoveries in Molecular, Cellular, and Evolutionary Biology, 288A 2: 191-204. doi:10.1002/ar.a.20282
Zhang, J. Y., Chen, Y. B., Hardwick, J. M., Miller, M. I., Plachez, C., Richards, L. J., Yarowsky, P., van Zijl, P. and Mori, S. (2005) Magnetic resonance diffusion tensor microimaging reveals a role for Bcl-x in brain development and homeostasis. Journal of Neuroscience, 25 8: 1881-1888. doi:10.1523/JNEUROSCI.4129-04.2005
Mapping postnatal mouse brain development with diffusion tensor microimaging
Zhang, J. Y., Miller, M. I., Plachez, C., Richards, L. J., Yarowsky, P., van Zijl, P. and Mori, S. (2005) Mapping postnatal mouse brain development with diffusion tensor microimaging. Neuroimage, 26 4: 1042-1051. doi:10.1016/j.neuroimage.2005.03.009
Mechanisms of axon guidance in the developing nervous system
Plachez, C. and Richards, L. J. (2005) Mechanisms of axon guidance in the developing nervous system. Current Topics In Developmental Biology, 69 267-346. doi:10.1016/S0070-2153(05)69010-2
The transcription factor gene Nfib is essential for both lung maturation and brain development
Steele-Perkins, George, Plachez, Céline, Butz, Kenneth G., Yang, Guanhu, Bachurski, Cindy J., Kinsman, Stephen L., Litwack, E. David, Richards, Linda J. and Gronostajski, Richard M. (2005) The transcription factor gene Nfib is essential for both lung maturation and brain development. Molecular and Cellular Biology, 25 2: 685-698. doi:10.1128/MCB.25.2.685-698.2005
A new chemotaxis assay shows the extreme sensitivity of axons to molecular gradients
Rosoff, W. J., Urbach, J. S., Esrick, M. A., McAllister, R. G., Richards, L. J. and Goodhill, G. J. (2004) A new chemotaxis assay shows the extreme sensitivity of axons to molecular gradients. Nature Neuroscience, 7 6: 678-682. doi:10.1038/nn1259
Mechanisms regulating the development of the corpus callosum and its agenesis in mouse and human
Richards, L. J., Plachez, C. and Ren, T. (2004) Mechanisms regulating the development of the corpus callosum and its agenesis in mouse and human. Clinical Genetics, 66 4: 276-289. doi:10.1111/j.1399-0004.2004.00354.x
Abnormal development of forebrain midline glia and commissural projections in Nfia knock-out mice
Shu, Tianzhi, Butz, Kenneth G., Plachez, Celine, Gronostajski, Richard M. and Richards, Linda J. (2003) Abnormal development of forebrain midline glia and commissural projections in Nfia knock-out mice. Journal of Neuroscience,23 1: 203-212.
Leingartner, A, Richards, LJ, Dyck, RH, Akazawa, C and O'Leary, DDM (2003) Cloning and cortical expression of rat Emx2 and adenovirus-mediated overexpression to assess its regulation of area-specific targeting of thalamocortical axons. Cerebral Cortex, 13 6: 648-660. doi:10.1093/cercor/13.6.648
Development of midline glial populations at the corticoseptal boundary
Shu, T. Z., Puche, A. C. and Richards, L. J. (2003) Development of midline glial populations at the corticoseptal boundary. Journal of Neurobiology, 57 1: 81-94. doi:10.1002/neu.10252
Neuropilin-1 conveys semaphorin and VEGF signaling during neural and cardiovascular development
Gu, C. H., Rodriguez, E. R., Reimert, D. V., Shu, T. Z., Fritzsch, B., Richards, L. J., Kolodkin, A. L. and Ginty, D. D. (2003) Neuropilin-1 conveys semaphorin and VEGF signaling during neural and cardiovascular development.Developmental Cell, 5 1: 45-57. doi:10.1016/S1534-5807(03)00169-2
Slit2 guides both precrossing and postcrossing callosal axons at the midline in vivo
Shu, T. Z., Sundaresan, V., McCarthy, M. M. and Richards, L. J. (2003) Slit2 guides both precrossing and postcrossing callosal axons at the midline in vivo. Journal of Neuroscience, 23 22: 8176-8184.
The glial sling is a migratory population of developing neurons
Shu, T. Z., Li, Y., Keller, A. and Richards, L. J. (2003) The glial sling is a migratory population of developing neurons.Development, 130 13: 2929-2937. doi:10.1242/dev.00514
Zhang, Jiangyang, Richards, Linda J, Yarowsky, Paul, Huang, Hao, van Zijl, Peter C. M and Mori, Susumu (2003) Three-dimensional anatomical characterization of the developing mouse brain by diffusion tensor microimaging.Neuroimage, 20 3: 1639-1648. doi:10.1016/S1053-8119(03)00410-5
Axonal pathfinding mechanisms at the cortical midline and in the development of the corpus callosum
Richards, L. J. (2002) Axonal pathfinding mechanisms at the cortical midline and in the development of the corpus callosum. Brazilian Journal of Medical And Biological Research, 35 12: 1431-1439. doi:10.1590/S0100-879X2002001200004
Surrounded by slit - How forebrain commissural axons can be led astray
Richards, LJ (2002) Surrounded by slit - How forebrain commissural axons can be led astray. Neuron, 33 2: 153-155. doi:10.1016/S0896-6273(02)00566-4
A role for cingulate pioneering axons in the development of the corpus callosum
Rash, B. G. and Richards, L. J. (2001) A role for cingulate pioneering axons in the development of the corpus callosum. Journal of Comparative Neurology, 434 2: 147-157. doi:10.1002/cne.1170
Cortical axon guidance by the glial wedge during the development of the corpus callosum
Shu, T. Z. and Richards, L. J. (2001) Cortical axon guidance by the glial wedge during the development of the corpus callosum. Journal of Neuroscience, 21 8: 2749-2758.
Shu, T. Z., Shen, W. B. and Richards, L. J. (2001) Development of the perforating pathway: An ipsilaterally projecting pathway between the medial septum/diagonal band of broca and the cingulate cortex that intersects the corpus callosum. Journal of Comparative Neurology, 436 4: 411-422. doi:10.1002/cne.1077
Adenoviral vector-mediated rescue of the OMP-null phenotype in vivo
Ivic, L, Pyrski, MM, Margolis, JW, Richards, LJ, Firestein, S and Margolis, FL (2000) Adenoviral vector-mediated rescue of the OMP-null phenotype in vivo. Nature Neuroscience, 3 11: 1113-1120.
Gad, JM, Keeling, SL, Shu, TZ, Richards, LJ and Cooper, HM (2000) The spatial and temporal expression patterns of netrin receptors, DCC and neogenin, in the developing mouse retina. Experimental Eye Research, 70 6: 711-722. doi:10.1006/exer.2000.0823
Shu, TZ, Valentino, KM, Seaman, G, Cooper, HM and Richards, LJ (2000) Expression of the netrin-1 receptor, deleted in colorectal cancer (DCC), is largely confined to projecting neurons in the developing forebrain. Journal of Comparative Neurology, 416 2: 201-212. doi:10.1002/(SICI)1096-9861(20000110)416:2<201::AID-CNE6>3.0.CO;2-Z
Retinotectal maps: molecules, models and misplaced data
Goodhill, GJ and Richards, LJ (1999) Retinotectal maps: molecules, models and misplaced data. Trends in Neurosciences, 22 12: 529-534. doi:10.1016/S0166-2236(99)01469-1
Retinal axon guidance by region-specific cues in diencephalon
Tuttle, R, Braisted, JE, Richards, LJ and O'Leary, DDM (1998) Retinal axon guidance by region-specific cues in diencephalon. Development, 125 5: 791-801.
Richards, LJ, Koester, SE, Tuttle, R and OLeary, DDM (1997) Directed growth of early cortical axons is influenced by a chemoattractant released from an intermediate target. Journal of Neuroscience, 17 7: 2445-2458.
Labeling neural cells using adenoviral gene transfer of membrane-targeted GFP
Moriyoshi, K, Richards, LJ, Akazawa, C, OLeary, DDM and Nakanishi, S (1996) Labeling neural cells using adenoviral gene transfer of membrane-targeted GFP. Neuron, 16 2: 255-260. doi:10.1016/S0896-6273(00)80044-6
Richards, LJ, Kilpatrick, TJ, Dutton, R, Tan, SS, Gearing, DP, Bartlett, PF and Murphy, M (1996) Leukaemia inhibitory factor or related factors promote the differentiation of neuronal and astrocytic precursors within the developing murine spinal cord. European Journal of Neuroscience, 8 2: 291-299. doi:10.1111/j.1460-9568.1996.tb01213.x
Lineage Specification of Neuronal Precursors in the Mouse Spinal-Cord
Richards, LJ, Murphy, M, Dutton, R, Kilpatrick, TJ, Puche, AC, Tan, SS, Talman, PS and Bartlett, PF (1995) Lineage Specification of Neuronal Precursors in the Mouse Spinal-Cord. Proceedings of the National Academy of Sciences of the United States of America, 92 22: 10079-10083. doi:10.1073/pnas.92.22.10079
The Regulation of Neural Precursor Cells Within the Mammalian Brain
Kilpatrick, TJ, Richards, LJ and Bartlett, PF (1995) The Regulation of Neural Precursor Cells Within the Mammalian Brain. Molecular and Cellular Neuroscience, 6 1: 2-15. doi:10.1006/mcne.1995.1002
Leukemia Inhibitory Factor Rescues Motoneurons From Axotomy-Induced Cell-Death
Cheema, SS, Richards, LJ, Murphy, M and Bartlett, PF (1994) Leukemia Inhibitory Factor Rescues Motoneurons From Axotomy-Induced Cell-Death. Neuroreport, 5 8: 989-992. doi:10.1097/00001756-199404000-00034
Cheema, SS, Richards, L, Murphy, M and Bartlett, PF (1994) Leukemia Inhibitory Factor Prevents the Death of Axotomised Sensory Neurons in the Dorsal-Root Ganglia of the Neonatal Rat. Journal of Neuroscience Research, 37 2: 213-218. doi:10.1002/jnr.490370207
Regulation of the Early Development of the Nervous-System by Growth-Factors
Bartlett, PF, Kilpatrick, TJ, Richards, LJ, Talman, PS and Murphy, M (1994) Regulation of the Early Development of the Nervous-System by Growth-Factors. Pharmacology & Therapeutics, 64 3: 371-393. doi:10.1016/0163-7258(94)00044-1
Rosenfeld, JV, Richards, LJ and Bartlett, PF (1993) Mutant Mouse Cerebellum Does Not Provide Specific Signals for the Selective Migration and Development of Transplanted Purkinje-Cells. Neuroscience Letters, 155 1: 19-23. doi:10.1016/0304-3940(93)90664-7
Richards, LJ, Kilpatrick, TJ, Bartlett, PF and Murphy, M (1992) Leukemia Inhibitory Factor Promotes the Neuronal Development of Spinal-Cord Precursors From the Neural-Tube. Journal of Neuroscience Research, 33 3: 476-484. doi:10.1002/jnr.490330314
Denovo Generation of Neuronal Cells From the Adult-Mouse Brain
Richards, LJ, Kilpatrick, TJ and Bartlett, PF (1992) Denovo Generation of Neuronal Cells From the Adult-Mouse Brain.Proceedings of the National Academy of Sciences of the United States of America, 89 18: 8591-8595. doi:10.1073/pnas.89.18.8591
Consortia Publications
Szczupak D., Yen C.C., Liu C., Tian X., Lent R., Tovar-Moll F. and Silva A.C. in collaboration with the IRC5 Consortium Dynamic interhemispheric desynchronization in marmosets and humans with disorders of the corpus callosum (2020). Frontiers in Neural Circuits doi:10.3389/fncir.2020.612595
International Brain Initiative (IBI) (2020) International Brain Initiative: An innovative framework for coordinated global brain research efforts. Neuron doi:10.1016/j.neuron.2020.01.002. * Richards L.J. co-corresponding author.
Erratum (2020) Neuron doi: 10.1016/j.neuron.2020.02.022.
A Neuroethics Framework for the Australian Brain Initiative
Australian Brain Alliance (2019) A Neuroethics Framework for the Australian Brain Initiative. Neuron doi: 10.1016/j.neuron.2019.01.004. * Richards L.J. co-corresponding author.
A Neuroethics Framework for the Australian Brain Initiative
Erratum (2020) A Neuroethics Framework for the Australian Brain Initiative. Neuron doi:10.1016/j.neuron.2019.12.019.
Australian Brain Alliance (2016) Australian Brain Alliance Neuron 92: 597-600. doi.org/10.1016/j.neuron.2016.10.038. *Richards L.J. corresponding author.
Responsible innovation in neurotechnology enterprises
Garden, H., et al. (2019) Responsible innovation in neurotechnology enterprises, OECD Science, Technology and Industry Working Papers, No. 2019/05, OECD Publishing, Paris, https://doi.org/10.1787/9685e4fd-en.
Techniques used in the lab include:
- Magnetic Resonance Imaging – diffusion tensor imaging (human and animal).
- In utero surgery, in utero electroporation of genetic constructs to label cells, over-express genes, or knock-down protein expression by siRNA.
- Tissue culture and in vitro primary cultures, including organotypic slice culture.
- Neuroanatomical and Histological approaches including tract tracing, immunohistochemistry, cryostat, vibratome, freezing microtome and electron and confocal microscopy
- Molecular Biological approaches including protein purification, PCR, Northern, and Western Blotting, in situ hybridisation, chromatin immunoprecipitation, luciferase assays and genomic analysis
Project 1: Human agenesis of the corpus callosum, autism spectrum disorder and brain wiring
Agenesis of the corpus callosum is a brain wiring alteration that occurs during brain development. Many people have some characteristics that are similar to those with autism spectrum disorder. We are investigating brain wiring connectivity using high-field magnetic resonance imaging and neuropsychological testing to understand how brain connectivity underpins the function of the brain. We also want to understand the underlying causes of agenesis of the corpus callosum by performing genetic analyses of DNA from people with these disorders compared to controls. The work will have a significant impact on our understanding of how changes in brain wiring impact brain function.
Opportunities exist for students with a background or interest in: Neuroscience, genetics, magnetic resonance imaging and physics, neuropsychology, medicine, computer science (data analysis and software development).
Project 2: Function of genes and molecules in agenesis of the corpus callosum and brain developmental disorders
Identifying a causal genetic mutation in a person requires functional studies to determine if the mutation causes a change in the function of the gene. This work requires in-depth analysis in animal models to examine gene function in cellular proliferation, differentiation, migration and cortical wiring. We are interested to understand the basic mechanisms regulating these developmental events and hwo they are altered in human brain disorders including agenesis of the corpus callosum, ventriculomegaly, hydrocephalus and cortical malformations. This work has a significant translational impact on understanding the causes of brain developmental disorders.
Opportunities exist for students with a background or interest in: Neuroscience, genetics, cell biology, developmental biology, glial development, animal behaviour, medicine.
Project 3: The function of early neuronal activity on the formation of neocortical circuits
How does the brain acquire its connectivity pattern during development? This project aims at elucidating the main roles of early sensory and spontaneous activity in the formation of neocortical circuits. By combining molecular, electrical and developmental manipulations in developing mammalian embryos and pups, this project will study how early events affect the precise formation of cortical features required for normal cognitive development. The work will have a significant impact on our understanding of how the brain is wired for function.
Opportunities exist for students with a background or interest in: Neuroscience, developmental neurobiology, neurophysiology, electrophysiological signal analysis and/or computational sciences, mathematical modelling, medicine.
Project 4: Principles of neural development applied to understanding brain cancer
Brain cancer is a significant health problem in Australia. One of the most aggressive forms of brain cancer is glioblastoma (GBM) and the prognosis for these patients is extremely poor. What is needed is a deeper understanding of the cause of brain cancer. We are approaching this challenge by utilising the principles of neural development to understand how tumours first arise in the brain and how they are able to continue to grow and metastasize in order to find the causes and treatments for adult and pediatric brain cancers that originate from glia. Nuclear factor one (NFI) genes have been implicated in brain cancer and in glial development. We have generated a number of animal models of Nfi gene mis-expression to determine the function of NFI genes in brain cancer. This work will have a significant impact on our understanding of the cause and progression of brain cancer.
Opportunities exist for students with a background or interest in: Neuroscience, genetics, cell biology, developmental biology, glial development, animal behaviour, medicine.
| Interview for Corpus Callosum Awareness Day | 01/07/2020 | Perth Now |
| Interview for Corpus Callosum Awareness Day | 01/07/2020 | Channel Nine News |
| Feature on the brain | 06/04/2020 | ABC 'Nightlife' Radio Program |
| 'Westside power list: our top academic minds' | 22/08/2019 | 'Westside News' |
|
‘It’s all in your head: understanding your brain’ |
18/02/2019 |
|
|
‘Same same but different: the evolution of the mammal brain’ |
04/09/2018 |
|
|
'Bridging the brain-mind gap’ |
09/05/2018 |
|
|
Article - ‘Out of their depth’ regarding the brains of teenagers |
09/2017 |
‘Courier Mail’ ‘QWeekend’ Magazine |
|
Comment on QBI researcher John McGrath’s latest psychosis and suicide risk research |
09/2017 |
Medical Journal of Australia |
|
‘Monday Expert’ – ‘Brain research fail: Scientists still have very little idea about what's going on up there' |
29/05/2017 |
|
|
Article - women in science |
03/2017 |
Westside News |
|
Interview - ‘The mysterious corpus callosum’ |
08/01/2017 |
|
|
Short article - research identifying protein which can affect growth of brain cancer |
07/2016 |
Australian Women’s Weekly |
|
Comment - disorders of the corpus callosum |
07/2016 |
Perth News |
|
Comment - disorders of the corpus callosum |
07/2016 |
Caboolture News |
|
Interview - ‘The mysterious corpus callosum’ |
16/05/2016 |
|
|
Interview - identification of a protein which plays a key role in brain cancer |
04/2016 |
Courier Mail |
|
Interview - ‘Cancer researchers find protein link that could boost detection and treatment’ |
20/04/2016 |
|
|
‘Top young brains compete in International Brain Bee Championship’ |
10/10/2015 |
|
|
Comment - International Brain Bee |
08/2015 |
AAP Newswire |
|
Article - 'Great Australian Spelling Bee gives way to Brain Bee in Queensland’ |
20/08/2015 |
|
|
‘Australian Brain Bee Challenge dispels myths, inspires students’ |
18/07/2015 |
|
|
‘A New Day Set to Raise Awareness of Central Brain Structure’ |
01/07/2015 |
|
|
Interview – ‘How sensory input can influence brain wiring’ |
08/2014 |
ABC Tropical North Radio |
|
iInterview - Launch of the 7T scanner and raising awareness of corpus callosum disorders |
06/2014 |
Channel 9 TV |
|
Interview - Raising awareness of ABBC and QBI |
06/2012 |
ABC TV 7.30 Report |
|
Interview - Raising awareness of ABBC and QBI |
03/2012 |
ABC 612 Radio |
|
Interview - Raising awareness of glioma research and QBI |
08/2011 |
Channel 9 TV |
|
Interview - Raising awareness of ABBC and QBI |
02/2010 |
ABC Sydney Radio |
|
Interview - Raising awareness of ABBC and QBI |
02/2010 |
Today Tonight TV |
|
Interview - ABBC |
03/2009 |
ABC 612 Radio
|
|
Interview - national final of the ABBC with Qld winner |
08/2008 |
ABC 612 Radio |
|
Interview - advertising brain awareness activities and raising the profile of brain research |
03/2007 |
Triple J Radio |
|
Interview - careers in neuroscience |
07/2007 |
Courier Mail |
|
Interview - activities for Brain Awareness Week at The University of Queensland |
03/2006 |
ABC 612 Radio |
|
Interview - advertising the Australian Brain Bee Challenge (ABBC) |
03/2006
|
ABC 612 Radio
|
|
Interview - ‘QBI recruits family of top neuroscientists’ |
01/2006
|
Westside News
|
|
Article - ABBC |
03/2006 |
Westside News |
|
Interview - issues related to my research work on agenesis of the corpus callosum as the Toshiya Yamada memorial Lecturer at the Institute for Molecular Bioscience |
03/2005 |
ABC 612 Radio
|
|
Interview - ABBC Mareeba Kids |
24/07/2014 |
ABC Far North Radio |
|
Interview - ABBC |
23/07/2014 |
4BC Radio |
|
‘Tannum team tops Australian Brain Bee Challenge’ |
08/07/2014 |
|
|
‘Brain Bee Challenge won by Queensland's 'brainiest' student Sophie Watson’ |
23/07/2014 |
|
|
‘Brain Bee Challenge won by Queensland's 'brainiest' student Sophie Watson’ |
22/07/2014 |
|
|
‘Brain Bee Challenge won by Queensland's 'brainiest' student Sophie Watson’ |
22/07/2014 |
Research Areas
- Axon guidance
- Cortical development
Latest news
-
-
Students head into the labs for Summer Research Program
17 December 2019